Did you add too much baking soda to your recipe and you're wondering what it's going to do to your baked goods? Find out everything you need to know about baking soda in baking: what it is, what it does, what happens if you add too much, if it expires, substitutions and what happens if you use baking soda instead of baking powder.

I shared with you a recipe for marmalade pudding cakes, pulled from my family's recipe box. The original recipe had a lot of baking soda, and the puddings browned significantly as they cooked. Like a good chemist, I blamed the baking soda and I wanted to investigate.

I remade the pudding cakes, modifying the amount of baking soda and adding only 0.625 mL (⅛ a teaspoon) to the second batch, instead of the full 5 mL (1 teaspoon) called for in the original recipe. The second batch of steamed puddings was completely different: significantly lighter in colour, with a firmer, spongy texture.
The only difference between the two cakes was the quantity of baking soda. Here's everything you need to know about baking soda, including what it is, what it does, how it works, how to store it, and how to replace it in baking.
Jump to:
What it is
Formula and structure
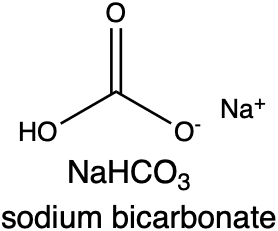
Baking soda is the common name for the chemical sodium bicarbonate (or bicarbonate of soda, as the Brits like to call it). The chemical formula for baking soda is NaHCO3. Sodium bicarbonate is basic (as in alkaline) and reacts under acidic conditions to produce carbon dioxide (CO2), the gas that lifts cake batters and helps your cakes rise (water and salt are also produced).
Primary use in baking
Sodium bicarbonate is a chemical leavener, unlike yeast. Yeast is a living organism, and when you feed it (like when you give it some water and some sugar), it too will produce gas, but through a different process known as fermentation: the yeast organisms eat sugar, digest it, and then they produce CO2 (along with other compounds like alcohol).
To summarize, your bread rises because the yeast in the bread dough eat sugar and produce gases, while your cakes rise because baking soda reacts with acids to produce a gas. This is also the principle behind baking powder, another leavening agent.
Now, why does more or less baking soda make the two batches of cake look so different? There's more chemistry to discuss here, like the Maillard reaction.

What it does
Baking soda reacts with vinegar and acids
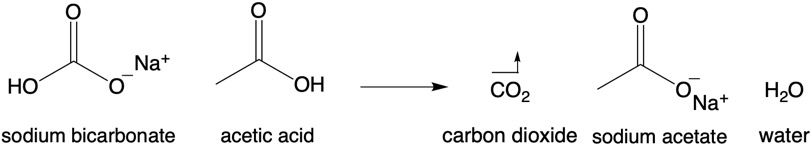
When baking soda is exposed to acid(s), it breaks down into carbon dioxide and water, as well as a salt. The salt that forms depends on the acid that it reacts with. For example, if baking soda reacts with vinegar: vinegar is made up of 5% acetic acid, so when the two react, carbon dioxide gas forms, as well as water and sodium acetate.
Browning of baked goods via the Maillard reaction
As you bake a cake or bread, you obviously notice a change in colour as the baked good turns golden brown, but you might also notice that the sweet flavours transform into something deeper, and not as sweet: French pastry chefs like to call this "golden brown delicious." Some of the browning you see comes from the caramelization of sugars (sugars break down/transform into brown-coloured polymers and aromatic substances that contribute to the aroma and flavour of baked goods).

Maillard browning reaction is also responsible for the colour and flavour changes you see. Maillard browning is faster under basic conditions: when the pH is above 7, the browning reactions are sped up. These browning reactions also depend on the presence of proteins. The compounds that form are different from the caramelization of sugars.
The Maillard reaction turns cakes from pale and very sweet to golden brown delicious. Baking soda and Maillard browning give Boston brown bread its signature taste and brown colour.

What happens if you add too much
There's a fine line between the right amount of baking soda and the wrong amount. Make sure to use the right methods to measure your ingredients.
Not sure if you need what leavening agent you need? Read about baking soda vs baking powder to better understand the differences!
Excessive browning
The Maillard reaction speeds up under basic conditions (like when you add to a recipe a lot of baking soda, which is alkaline, i.e. basic). Because the original marmalade pudding recipe had a large amount of baking soda in it, the resulting cake batter had a higher pH, and the Maillard reaction occurred faster. The puddings browned more quickly while steaming for an hour. More baking soda, more browning.
Odd soapy taste
Unfortunately, with more baking soda, a lot of it reacts, but some of it (the excess) is left behind, unreacted. This lingering baking soda affects the flavour, which seems "sharper" and too much baking soda might cause your cakes and cookies to taste soapy even.

The puddings made with a full teaspoon of baking soda taste less like marmalade and more like something stronger than the expected citrus flavour. That's the baking soda. The second batch of puddings had only an eighth of a teaspoon of baking soda, therefore the Maillard reaction occurred much slower. The steamed puddings have a more familiar "golden brown delicious" look to them. The taste of the marmalade is clear, and so is the delicious buttery flavour.
At this point, you would think I'm done with the chemistry chat, but I'm not. I have one more thing to point out about baking soda.
Tenderizing effect and impact on texture
I noticed the original pudding cakes had a very tender crumb, and they were spongy and soft. On the other hand, the puddings made with one-eighth of the baking soda were still spongy, but much firmer. Once again, we can blame baking soda. Baking soda provides lots of rising power to the pudding cakes, but actually, I noticed that the cakes with less baking soda were more domed, while the cakes with more baking soda were flatter, but with a more bubbled texture on the sides and bottom.
The baking soda raised the pH of the cake batter, thereby weakening the gluten in the flour: the texture and tenderness of the cakes were affected. Weaker gluten means a looser structure, with more spread, bigger air pockets (a more open crumb) and tenderness. So the cakes with more baking soda seemed to rise less, but in fact, that's because the gluten was weak and couldn't support the forming gas pockets. An adequate amount of baking soda (⅛ tsp) allowed the little pudding cakes to rise up and stay up, while an excess of baking soda caused the cakes to spread out, instead of up.
How much to use in cakes and other recipes-ratio and rule of thumb
Too much baking soda is clearly not a good thing, creating too many bubbles in cakes, causing cakes to sink, leading to over-browning, and producing an off-flavour that might even be soapy. So how much baking soda is enough?
The basic rule for how much baking soda to add to a recipe is 1.25 mL (¼ teaspoon) of baking soda for every 125 grams (1 cup or 250 mL) of all-purpose flour.
In recipes where that contain large amounts of acidic ingredients, you would need 2.5 mL (½ teaspoon) of baking soda to neutralize 1 cup of a mildly acidic ingredient, like sour cream, buttermilk, or yogurt. For example, this Irish soda bread with raisins is made with 500 mL (2 cups) of buttermilk and 1 teaspoon of baking soda. Of course, this is just a guideline and recipes may vary for other reasons (pan size, presence of "heavy" ingredients like nuts and dried fruit that might require extra leavening, etc.).
Shelf life, storage, and stability
Baking powder does expire and can lose potency over time, given that baking powder contains both baking soda and an acid (or two) that it can react with, in presence of humidity. For this reason, it is super important to check baking powder periodically to make sure it is still reactive. On the other hand, baking soda is just sodium bicarbonate, a single compound and even if it's exposed to a little humidity from the air, it won't lose potency the same way baking powder does and it takes much longer. In fact, baking soda is considered to be shelf stable for at least 2 years when stored under typical room temperature conditions (25 °C with 60 % humidity). For this reason, baking soda does not expire.
The problem with older containers
Though baking soda doesn't expire, you still have to be weary when baking with a container of baking soda that has been open for months (or even years). It's not that the baking soda will break down over time and stop working, but open containers of baking soda exposed to humidity will clump. You will have trouble incorporating clumpy baking soda into your cake batters and other baked goods, which can lead to clusters of baking soda in cake batters and cookie doughs. These clusters will lead to tiny patches of baking soda in your baked goods, causing brown spots and larger air pockets in your baked goods.
Baking soda does break down very slowly over time, but more rapidly when heated (above 140°F/60°C) and forms sodium carbonate, water, and carbon dioxide. Some bakers will heat dry baking soda on a sheet pan in the oven with the goal of forming sodium carbonate. They will then use the sodium carbonate to boil pretzels before baking to promote darker browning on the crust (from Maillard browning). Bathing pretzels in a bath of either sodium carbonate or sodium bicarbonate is a pretty decent way to make dark brown pretzels without having to use sodium hydroxide which is more corrosive and a little harder to come by.
Storage recommendations
Given that baking soda will form clumps if exposed to moisture, it's important to store it in a cool, dry place, in a closed container that has a good seal to make sure moisture doesn't get in. Baking soda is often sold at the grocery store in a cardboard box that, once opened, doesn't shut, so you may want to either store the box in a bag with a seal or transfer the powder to an air-tight container with a proper lid. This way air exposure will be minimal and your baking soda won't clump.
Substitutions
If you don't have baking soda, replacing it in a recipe depends on what you are trying to make:
- In cakes, replace ¼ teaspoon of baking soda with 1 teaspoon of baking powder and replace all or a portion of acidic ingredients (like buttermilk) with regular milk, which has a higher pH. This banana bread without baking soda is made with baking powder instead and relies on the acidity of the bananas (and it works!)
- In muffins, replace ¼ teaspoon of baking soda with 1 teaspoon of baking powder and replace all or a portion of acidic acidic ingredients (like
- In cookies, you can't easily replace baking soda. It's a little more complicated because baking soda helps cookies spread when they bake and also contributes to browning by raising the pH of the dough. Baking powder is not a good replacement for baking soda in most cookie recipes.
- buttermilk) with regular milk, which has a higher pH
- Exception: in snickerdoodle cookies, you could replace baking soda plus cream of tartar with baking powder. These chocolate snickerdoodle cookies can actually be made both ways!
If you are avoiding sodium in your diet, you may be looking for a baking soda substitute. In this case, consider baking with potassium bicarbonate: you can replace baking soda with the same amount of potassium bicarbonate. It is more expensive and harder to find. It's not readily available in most grocery stores.
Frequently asked questions
You can replace the baking powder with baking soda in most recipes, as long as there's an acidic ingredient present in the list of ingredients. If not, you will have to also introduce an acid in your recipes, such as cream of tartar or citric acid (which are dry powders and would be suitable in cookie recipes with baking powder), or lemon juice, buttermilk, yogurt, or sour cream (which are liquids and therefore more appropriate for cakes and muffins).
Neither one is better and they have overlapping roles. Which you choose to use will depend on your recipes and your goals. In a cookie recipe, baking soda may be more appropriate to promote faster browning in a short baking time and also to help cookies spread out a little by weakening the gluten structure. In cakes, baking powder is more convenient because it is a complete leavening agent that will react regardless of the pH and the other ingredients present in your recipe.
Replacing baking soda with baking powder in a cookie recipe will change the texture of the cookie, the colour, and also the thickness. Cookies made with baking powder may brown less in the oven, may be thicker and rise taller, and spread less. Cookies made with baking soda may spread out more, be thinner, and bake to a deeper golden brown colour.
Baking soda is pure sodium bicarbonate and therefore is aluminum-free. It does not contain aluminum.
Conclusion
Baking soda is an important leavening agent in baking. We all use it and it is a vital ingredient in commercial and homemade baking powder. But actually, baking soda does so much more than just cause cakes to rise. Baking soda is alkaline, and as such, adding it to recipes means that it may affect the colour, flavour, and texture of baked goods, as does too much baking powder.







Mallory Frayn says
Not boring at all, I love science! I cannot believe how drastic the colour difference was though.
Janice Lawandi says
I know! Isn't it amazing! And it's the exact same "steam time" in both cases. I even asked my mom about the darker color, and she says that her mom's pudding cakes were always this dark. It's really the baking soda!
Franceska says
I just loved this post! Please keep posting things like this! It's really interesting 🙂
Janice Lawandi says
I will try! I just have to come up with more topics and examples 😉
Christelle is flabbergasting says
You didn't bore me at all! So interesting! So, which one tasted the best for you? (at 1/8 tsp?)
Janice Lawandi says
I'm glad I didn't bore you 😉
I definitely preferred the flavour of the puddings made with 1/8 tsp baking soda. They had a better butter flavour and the marmalade was more recognizable. I'd definitely use 1/8 tsp baking soda next time!
Mardi Michels says
Janice - I LOVE this post! Please make the "chemistry in the kitchen" a regular feature!
Christelle is flabbergasting says
I AGREE with Mardi!
Janice Lawandi says
Okay, okay 😉
And thank you for the encouragement!
Jerlyn says
Hi Janice,
Thank you so much for this awesome experiment with baking soda. I'm glad I came up to page. Very informative. I am new to baking and so I wanted to curious to know what each ingredient does in a recipe. I hope to read more baking experiments from you. Have a nice day!
bellwilde says
Thank you for the education. I was curious which tasted the best to you?
Janice Lawandi says
Honestly, I like the cakes with 1/8 tsp baking soda better (sorry family recipe!). The flavour of the marmalade was much more prominent and so was the rich butter taste in the puddings with 1/8 tsp baking soda!
wannafoodie says
Awesome post, especially the visual/side by side comparison!
Liz says
Really interesting blog, Janice!
Isabelle @ Crumb says
Thanks for taking us for a walk on the nerdy side. 🙂 I've always been intrigued by the chemistry of baking... after years in the kitchen, I have a pretty good idea of what happens when I adjust the amounts of certain ingredients, but I couldn't tell you why it happens. Seriously, love everything about this post. Please write more!
Adrienne says
Super interesting! Great post. So well written! Such a good read.
Tora says
This was incredibly interesting, thank you so much for sharing! I have attempted to caramelize onions faster by adding baking soda. It sort of worked, but I think I got too excited and impatient and added way too much of it so the onion just disintegrated. Now I shall explore more of this wonder that is baking soda! Thank you.
Rosr says
I have a recipe for chocolate sour cream pound cake. The ingredients listed 1/2 teaspoon baking powder but the instructions says add baking powder and baking soda but does say how much baking soda. Help please 🤗